Harvard Introduces a Swifter Way for 3D-Printed Organs
September 11, 2019
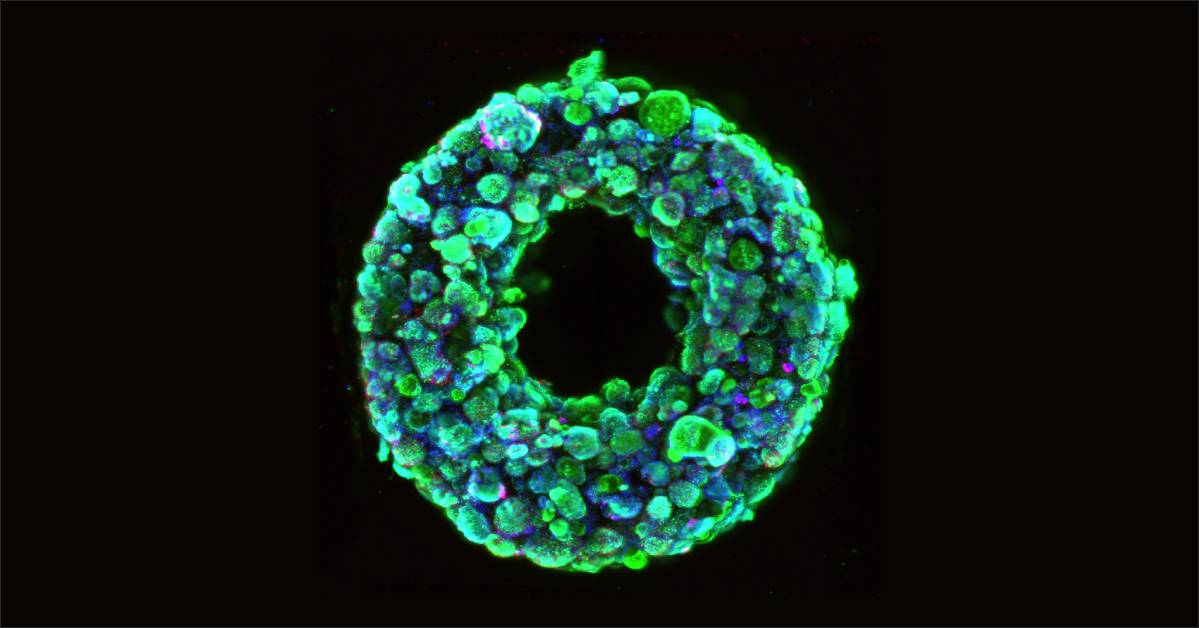
The researchers at Wyss Institute for Biologically Inspired Engineering of Harvard and John A. Paulson School of Engineering and Applied Sciences has created a new technique SWIFT (sacrificial writing into functional tissue) that overcomes major hurdles by 3D bioprinting vascular channels into living matrices composed of stem-cell derived organ building blocks (OBBs), yielding viable, organ-specific tissues with high cell density and function.
“This is an entirely new paradigm for tissue fabrication,” said co-first author Mark Skylar-Scott, Ph.D., a Research Associate at the Wyss Institute. “Rather than trying to 3D-print an entire organ’s worth of cells, SWIFT focuses on only printing the vessels necessary to support a living tissue construct that contains large quantities of OBBs, which may ultimately be used therapeutically to repair and replace human organs with lab-grown versions containing patient’s new cells.”
There are two steps involved in the process of SWIFT. It starts with forming hundreds of thousands of stem-cell-derived aggregates into a dense, living matrix of OBBs that contains about 200 million cells per millilitre, followed by the next step where a vascular network through which oxygen and other nutrients can be delivered to the cells is embedded with the matrix by writing and removing a sacrificial ink.
“Forming a dense matrix from these OBBs kills two birds with one stone: not only it achieve a high cellular density akin to that of human organs, but the matrix’s viscosity also enables printing of a pervasive network of perfusable channels within it to mimic the blood vessels that support human organs,” said co-first author Sebastien Uzel, Ph.D., a Research Associate at the Wyss Institute and SEAS.
The cellular aggregates used in the SWIFT method are derived from adult induced pluripotent stem cells, which are mixed with a tailored extracellular matrix (ECM) solution to make a living matrix that is compacted via centrifugation.
Organ-specific tissues, printed with embedded vascular channels using SWIFT remained viable, while tissues grown without these channels experienced cell death in their cores within 12 hours.
“Our SWIFT biomanufacturing method is highly effective at creating organ-specific tissues at scale from OBBs ranging from aggregates of primary cells to stem-cell-derived organoids,” said corresponding author Jennifer Lewis, Sc.D., who is a Core Faculty Member at the Wyss Institute as well as the Hansjorg Wyss Professor of Biologically Inspired Engineering at SEAS. “By integrating recent advances from stem-cell researchers with the bioprinting methods developed by my lab, we believe SWIFT will greatly advance the field of organ engineering around the world.”
There has been a number of collaborations going on with Wyss Institute faculty members Chris Chen, M.D., Ph.D. at Boston University and Sangeeta Bhatia, M.D., Ph.D., at MIT to implant these tissues into animal models and explore their host integration, as part of the 3D Organ Engineering Initiative co-led by Lewis and Chris Chen.
“The ability to support living human tissues with vascular channels is a huge step toward the goal of creating functional human organs of the body,” said Wyss Institute Founding Director Donald Ingber, M.D., Ph.D., who is also the Judah Folkman Professor of Vascular Biology at HMS, the Vascular Biology Program at Boston Children’s Hospital, and Professor of Bioengineering at SEAS. “We continue to be impressed by the achievements in Jennifer’s lab including this research, which ultimately has the potential to dramatically improve both organ engineering and the lifespans of patients whose own organs are failing.”
Source: Wyss. Harvard